Selective and Controllable Defluorophosphination and Defluorophosphorylation of Trifluoromethylated Enones: An Auxiliary Function of the Carbonyl Group
Li-Wen Sun,a Ya-Fei Hu,a Wen-Jun Ji,a Peng-Yuan Zhang,a Mengtao Ma,b Zhi-Liang Shen*a and Xue-Qiang Chu*a
a Technical Institute of Fluorochemistry (TIF), Institute of Advanced Synthesis, School of Chemistry and Molecular Engineering, Nanjing Tech University, Nanjing 211816, China. E-mails: ias_zlshen@njtech.edu.cn; xueqiangchu@njtech.edu.cn.
b Department of Chemistry and Materials Science, College of Science, Nanjing Forestry University, Nanjing 210037, China.
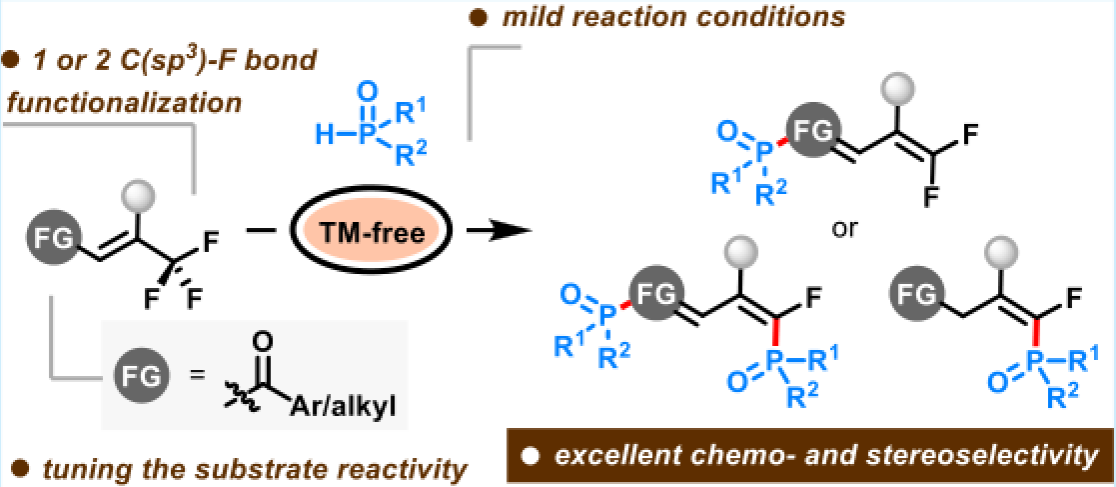
Abstract: The auxiliary function of a carbonyl group in the tunable defluorophosphination and defluorophosphorylation of trifluoromethylated enones with P(O)-containing compounds was demonstrated. Controlled replacement of one or two fluorine atoms in trifluoromethylated enones while maintaining high chemo- and stereoselectivity was achieved under mild conditions, thus enabling diversity-oriented synthesis of skeletally diverse organophosphorus libraries (Z)-difluoro-1,3-dien-1-yl phosphi-nates, (1Z,3E)-4-phosphoryl-4-fluoro-buta-1,3-dien-1-yl phosphi-nates, and (E)-4-phosphoryl-4-fluoro-1,3-but-3-en-1-ones in good yields with excellent functional group tolerance.
Organic Letters 2023, 25, 3745-3749. (Impact factor: 5.2)
论文链接:https://doi.org/10.1021/acs.orglett.3c01215
